Radiation-induced dysphagia is a significant complication for patients receiving radiotherapy for head and neck cancer, substantially affecting long-term quality of life. Despite advancements in concurrent chemoradiotherapy and intensity-modulated radiotherapy (IMRT), challenges persist. This narrative review synthesizes current knowledge about radiation-induced dysphagia, addresses clinical challenges, and highlights strategies to mitigate swallowing dysfunction post-radiotherapy. We conducted a comprehensive literature review focusing on the pathophysiology of dysphagia, critical structures related to dysphagia and aspiration, and radiological assessment techniques, including magnetic resonance imaging and computed tomography. This review also compares advancements in dosimetric optimization, such as dysphagia-optimized IMRT (Do-IMRT) and proton therapy, with conventional radiotherapy techniques. Radiation-induced dysphagia originates from progressive fibrosis, neuromuscular dysfunction, and direct damage to swallowing structures, especially the pharyngeal constrictors, cricopharyngeus, and larynx. Dose-volume relationships demonstrate a clear correlation between the severity of dysphagia and radiation exposure to the middle and inferior pharyngeal constrictors and larynx. Studies indicate that Do-IMRT significantly lessens long-term dysphagia by reducing radiation exposure to critical swallowing structures. Furthermore, early implementation of targeted rehabilitation interventions during and post-radiotherapy enhances swallowing function and improves quality of life. The advancement of precision radiation techniques, anatomical contouring, and proactive rehabilitation strategies have markedly enhanced the management of radiation-induced dysphagia without compromising oncologic outcomes. Do-IMRT represents a transformative approach, optimizing functional outcomes while reducing late toxicity. Future research should aim to refine personalized radiotherapy strategies and integrate comprehensive multidisciplinary rehabilitation protocols to further enhance swallowing outcomes in patients undergoing radiotherapy for head and neck cancer.
Head and neck cancer ranks as the seventh most common cancer globally, with approximately 669,000 new cases and 325,000 deaths annually [1]. This significant disease burden highlights the necessity for effective treatment strategies that balance oncologic control with functional preservation. Radical radiotherapy, combined with concurrent chemotherapy, remains the standard approach for advanced-stage disease, providing a viable organ-preserving alternative to surgery [2]. Extensive randomized studies and meta-analyses have confirmed that this strategy achieves survival outcomes comparable to those of radical resection, reinforcing its role as a frontline treatment [3].
Quality of Life Considerations
Beyond mere survival, it is essential to evaluate treatment outcomes in terms of post-therapy quality of life. The functional integrity of preserved organs significantly impacts long-term patient well-being and determines the overall success of organ preservation strategies [4]. Xerostomia and dysphagia rank as major post-treatment complications and are crucial determinants of diminished quality of life [5]. Although radiation-induced xerostomia has traditionally been viewed as the primary contributor to morbidity, recent evidence indicates that swallowing dysfunction has a more substantial impact. Langedijk et al. have documented that dysphagia significantly affects quality of life more than xerostomia, especially in the first 18 months after completing radiotherapy [6].
Advancements in Radiation Therapy
The introduction of modern radiation techniques, such as intensity-modulated radiotherapy (IMRT) with image guidance, has significantly enhanced organ preservation by sparing critical structures involved in swallowing. Specifically, minimizing radiation exposure to dysphagia and aspiration-related structures (DARS) has emerged as a strategic approach to mitigate post-treatment swallowing impairments and improve quality of life [7]. As survival rates increase and access to conformal radiotherapy becomes more widespread, optimizing dose constraints for DARS has become a crucial aspect of clinical practice.
Challenges in Dysphagia-Optimized IMRT
Despite its potential benefits, the implementation of dysphagia-optimized IMRT (Do-IMRT) presents significant challenges. The anatomical proximity of DARS to tumor sites, particularly in pharyngeal and laryngeal cancers, complicates the precise reduction of dose without compromising oncologic efficacy. Achieving a balance between effective tumor eradication and functional preservation is a fundamental challenge in treatment planning, requiring sophisticated contouring techniques and advanced dosimetric strategies.
Review Objectives
Achieving a comprehensive understanding of dysphagia following head and neck radiotherapy necessitates a multidisciplinary approach. This review integrates insights from pathophysiology, radiological assessment, contouring methodologies, and dose optimization strategies for DARS. Additionally, it emphasizes the importance of standardized assessment tools and evidence-based rehabilitation interventions to enhance post-treatment swallowing function. By synthesizing current knowledge in these areas, the review provides a detailed perspective for oncologists, radiologists, pathologists, and caregivers. Its goal is to address the morbidity associated with radiation-induced dysphagia and refine clinical strategies, thereby improving functional outcomes and enhancing long-term quality of life for head and neck cancer patients undergoing radiotherapy.
Prior to exploring the effects of radiation, it is essential to clearly define the normal swallowing mechanism. Swallowing is an intricately coordinated neuromuscular activity that plays a crucial role in the efficient and safe transport of food, while simultaneously protecting the airway. This complex activity is organized into three distinct phases, each requiring precise synchronization of cranial nerves, masticatory muscles, pharyngeal constrictors, and laryngeal structures. These elements are fundamental in driving the bolus from the oral cavity to the upper esophageal sphincter, ensuring effective airway protection. A thorough comprehension of the anatomical, physiological, and biomechanical aspects is vital for discerning the pathophysiological changes induced by radiation, which is critical for accurate diagnosis and management.
Preparatory Oral Phase
Food is chewed and mixed with saliva to form a cohesive bolus. This phase involves the coordinated activity of the jaw, tongue, and orofacial muscles, crucial for adequate mastication and bolus formation.
Transition to Oral Phase
Following preparation, the oral phase begins as the bolus is compressed against the hard palate by the dorsal surface of the tongue and propelled posteriorly by tongue base contraction. This voluntary action marks the shift from passive food processing to active transport toward the pharynx.
Pharyngeal and Esophageal Phases
As the bolus enters the pharynx, it is temporarily held in the vallecula, ensuring airway protection. The epiglottis tilts posteriorly to shield the laryngeal inlet, preventing aspiration. Concurrently, the sequential contraction of the pharyngeal constrictors propels the bolus toward the relaxed upper esophageal sphincter, facilitating its passage into the esophagus.
Clinical Implications
The phases of swallowing demonstrate the complexity and susceptibility of this mechanism to disruptions. Radiation-induced changes can significantly impair this coordination, resulting in dysfunction that carries profound clinical consequences. The subsequent section examines how these alterations present in patients with head and neck cancer. It provides a systematic framework to assess the impact of dysphagia and to delineate effective management strategies.
Building upon the fundamentals of the normal swallowing process, this section examines how dysphagia develops specifically as a result of radiation therapy in head and neck cancer treatments. It highlights the significant alterations to swallowing functions caused by radiation-induced changes and their treatment-related complications.
Dysphagia, characterized by difficulty in swallowing, arises from structural or functional abnormalities that can occur anywhere from the lips to the gastric cardia. Among patients with advanced head and neck cancer, swallowing dysfunction frequently manifests as a significant complication directly related to the effects of radiation therapy, irrespective of the specific chemoradiotherapy protocols employed.
Predisposing Factors for Dysphagia
Multiple factors contribute to dysphagia in this patient group. Although tumor progression can directly impair swallowing, dysphagia more often stems from residual damage to DARS and neuromuscular dysfunction following extensive tumor reduction. Xerostomia, a prevalent side effect of radiotherapy, further exacerbates swallowing difficulties, particularly with solid foods. Additional systemic factors such as general debilitation, pain, impaired pulmonary function, severe nausea, and gastroesophageal reflux also significantly impair swallowing function.
Radiation Effects on Swallowing
Radiation-induced dysphagia arises primarily from direct tissue damage and progressive fibrosis affecting DARS. These pathological changes impair bolus propulsion, disrupt the coordination of pharyngeal constrictors, diminish laryngeal elevation, and delay the relaxation of the upper esophageal sphincter. The collective impact of these alterations hampers bolus transit, leads to residue accumulation in the pharyngeal recesses, and elevates the risk of aspiration. These disturbances underscore the profound impact of radiation on swallowing function
Clinical Implications of Radiation-Induced Dysphagia
Dysphagia in head and neck cancer arises from a complex interplay of tumor burden, treatment-related tissue injury, and neuromuscular dysfunction. Radiation damages critical swallowing structures, exacerbated by xerostomia and systemic decline, markedly reducing swallowing efficiency and increasing aspiration risk. These impairments compromise nutrition, quality of life, and treatment adherence, necessitating early detection and customized management approaches.
Radiotherapy leads to a spectrum of functional impairments in the swallowing mechanism, many of which have been quantitatively characterized through instrumental assessments. Notably, videofluoroscopy studies have revealed profound alterations in swallowing physiology post-treatment [8]. These assessments show a marked reduction in pharyngeal peristalsis, often accompanied by impaired synchronization of pharyngeal contractions. Additionally, the diminished posterior inversion of the tongue base significantly reduces bolus propulsion. Compromised laryngeal closure, frequently observed as incomplete or delayed, substantially increases the risk of aspiration.
The impact of radiotherapy extends to hyoid bone elevation, a critical component for efficient swallowing mechanics. Decreased hyoid elevation compromises bolus clearance and may exacerbate dysphagia. Moreover, delayed opening of the upper esophageal sphincter further impedes bolus transit and clearance, complicating recovery and management [8].
Beyond these objective physiological changes, many patients report persistent subjective symptoms that profoundly affect daily functioning and overall quality of life. Common issues include difficulties with eating, necessary dietary modifications, social withdrawal during meals, and anxiety about choking [4,5]. These symptoms frequently extend beyond the acute treatment phase and are intricately linked to long-term treatment-related toxicities [6]. The integration of both objective assessments and patient-reported outcomes is crucial to fully comprehend the impact of radiotherapy-induced dysphagia and to facilitate comprehensive, patient-centered care [4–6].
Following the functional deficits previously discussed, radiotherapy induces a range of histopathological changes that progressively impair swallowing. These tissue-level changes are often subtle during initial clinical assessments but are pivotal in driving the chronic complications associated with radiation-induced dysphagia. Key mechanisms involved include persistent inflammation, progressive fibrosis, microvascular injury, muscle fiber atrophy, collagen deposition, and neural involvement. Collectively, these factors lead to the long-term structural deterioration of swallowing-related anatomy, setting the stage for the specific pathological manifestations that follow.
Late Radiation-Induced Mucositis
Characterized by mucosal discoloration, thinning, rigidity, and induration of subcutaneous tissues, late radiation-induced mucositis can escalate to ulceration and necrosis in severe cases. These changes primarily stem from chronic ischemia, which is a direct result of progressive fibrosis and small vessel occlusion. Beyond being a mucosal complication, this condition significantly impacts swallowing. The rigidity and ulceration of the mucosa, along with submucosal changes, can impair sensory inputs, delay swallowing reflexes, and obstruct bolus transit. These effects exacerbate dysphagia, heighten aspiration risks, and diminish oral intake, thereby severely impacting nutrition and quality of life [6].
Muscular Involvement in Dysphagia
Radiation significantly affects the pharyngeal constrictors, the primary muscles involved in swallowing. These muscles show an increase in thickness from approximately 2 mm before treatment to 7 mm after radiotherapy [7]. Radiation also impacts the pterygoid and masseter muscles, often leading to trismus due to cartilage thinning and synovial fluid depletion, which contributes to temporomandibular joint dysfunction [9]. Moreover, the dysfunction of the cricopharyngeus muscle further reduces swallowing efficiency [10].
Neural Impairment and Swallowing Dysfunction
The extent of dysfunction in pharyngeal constrictors, whether due to direct muscle damage or neural impairment, is not fully understood. The pharyngeal plexus provides terminal innervation to the pharynx, receiving inputs primarily from cranial nerves V, VII, IX, and X. These nerves pass through the connective tissue sheath that surrounds the pharyngeal muscles, extending into the muscle layer, mucosa, and submucosa. Radiation-induced mucosal damage may disrupt both afferent and efferent neural pathways, leading to sensory and motor deficits that further compound dysphagia [10].
Histological Basis of Radiation-Induced Dysphagia
Radiotherapy for head and neck cancer initiates a series of histopathological changes that chronically progress to dysphagia. The treatment triggers persistent inflammation, fibrosis, microvascular injury, muscle atrophy, and neural disruption. Collectively, these alterations impair bolus transit and compromise airway protection, elucidating the complex pathogenesis of dysphagia and highlighting the necessity of integrating histological insights into treatment planning.
Radiotherapy not only causes functional impairments and histopathological alterations but also induces significant radiological changes. These changes are crucial for understanding the structural basis of dysphagia. Advanced imaging techniques such as magnetic resonance imaging (MRI) and computed tomography (CT) play essential roles in the post-treatment surveillance of head and neck cancer patients. Although primarily used to monitor tumor responses and detect recurrences, the value of these modalities in identifying chronic complications like dysphagia is often overlooked in clinical practice.
MRI for Muscle Pathology Detection
Contrast-enhanced MRI of the face and neck is a standard follow-up procedure in both definitive and adjuvant radiotherapy settings. Radiological assessments typically focus on oncologic endpoints, evaluating primary tumors and cervical lymph nodes, and often neglect changes in nonmalignant tissues. This oversight can obscure the significant benefits of MRI in detecting changes in muscles that significantly impact swallowing functionality.
MRI is particularly adept at revealing soft tissue changes, enabling the detailed visualization of mesenchymal transformations including edema, inflammation, fat infiltration, myositis, and fibrosis. Notably, muscle edema, which can manifest even in the initial stages of radiation-induced dysphagia, is readily identifiable with MRI [11].
Dose-Dependent MRI Muscle Changes
MRI has proven instrumental in elucidating the dose-dependent effects of radiation on muscle tissue, as demonstrated by the research conducted by Popovtzer et al. [12]. This study assessed variations in T1- and T2-weighted signal intensities alongside changes in muscle thickness among 12 patients diagnosed with head and neck cancer. MRI evaluations were carried out before the onset of radiotherapy and again three months post-treatment.
The investigation focused on two critical muscle groups. The pharyngeal constrictors, essential for swallowing, were the primary subject of analysis. Additionally, the sternocleidomastoid muscles, though not directly involved in swallowing, were examined due to their substantial exposure to radiation owing to their location.
Three months after completing radiotherapy, notable changes were observed in both muscle groups. There was a decrease in T1-weighted signal intensity and an increase in T2-weighted signal intensity, with these changes closely tied to the dosage of radiation received. More precisely, T1 signal intensity diminished significantly in both the pharyngeal constrictors (p = 0.03) and the sternocleidomastoid muscles (p = 0.003) at doses exceeding 50 Grays (Gy). Conversely, T2 signal intensity escalated by 200% in the pharyngeal constrictors and 50% in the sternocleidomastoid muscles, underscoring a dose-responsive relationship.
The variation in muscle thickness also demonstrated dose dependency. In the pharyngeal constrictors, thickness increased by 66% among patients who received radiation doses under 50 Gy, compared to a more pronounced increase of 118% in those subjected to higher doses (p = 0.02). In stark contrast, thickness in the sternocleidomastoid muscles significantly decreased following treatment (p = 0.002).
These morphological changes imply that radiation-induced edema is a pivotal factor in the onset of dysphagia. Characterized radiographically by T1 hypointensity and T2 hyperintensity, this edema reflects the acute inflammatory responses occurring within the irradiated muscle tissue, illustrating the profound impact of radiotherapy on muscular structure and function.
CT-Based Evaluation of Pharyngeal Constrictors
CT also facilitates the assessment of muscular changes after radiotherapy. In a prospective study involving 26 patients, contrast-enhanced CT scans were performed before and three months after treatment to evaluate the thickness of pharyngeal constrictor muscles. Measurements were taken at three axial levels corresponding to the superior, middle, and inferior pharyngeal constrictors: the mid-C2 vertebral body, the inferior border of the hyoid bone, and the mid-cricoid cartilage.
Following radiotherapy, the median thickness of the pharyngeal constrictors increased from 2.5 mm to 7 mm (p = 0.001). Among patients without laryngeal tumors, the median midline thickness of the supraglottic larynx increased from 2 mm to 4 mm (p < 0.001). However, no significant changes were noted in other muscle groups involved in swallowing, including the suprahyoid muscles, longitudinal pharyngeal muscles, tongue surfaces, and the base of the tongue [7].
Radiologic Insights into Dysphagia Pathogenesis
Radiologic imaging provides crucial insights into the structural changes that underpin radiation-induced dysphagia. MRI and CT scans specifically uncover dose-related alterations in pharyngeal muscles, such as increased thickness, signal changes, edema, and fibrosis. These changes are directly correlated with swallowing impairments but are frequently overlooked in post-treatment evaluations.
Building on the detailed examination of functional, histological, and radiological changes previously outlined, it is essential to further investigate the anatomical structures most susceptible to radiation-induced damage and their contribution to dysphagia. Swallowing is an exceptionally complex physiological function that depends on the synchronized activity of over 30 muscle pairs and six cranial nerves. These structures collectively facilitate efficient bolus transit and ensure airway protection. Given their widespread anatomical distribution, it is technically impracticable to limit radiation exposure to all these structures without affecting target volume coverage.
Therefore, modern radiotherapy planning emphasizes a selective approach. This method prioritizes sparing the most crucial regions, collectively known as DARS. This strategy is pivotal in minimizing treatment-related morbidity while ensuring oncological effectiveness.
Key Structures in Radiation-Induced Dysphagia
Transitioning from strategic considerations to specific anatomical details, the structures most closely associated with post-radiotherapy dysphagia and aspiration have been clearly identified through videofluoroscopic evaluations [13]. Common abnormalities observed include reduced pharyngeal peristalsis and poor synchronization among pharyngeal contraction, upper esophageal sphincter opening, and laryngeal closure. The primary anatomical contributors implicated are the circular pharyngeal constrictors; longitudinal pharyngeal wall muscles such as stylopharyngeus, salpingopharyngeus, and palatopharyngeus; glottic and supraglottic laryngeal structures along with their intrinsic adductor muscles; suprahyoid muscles including geniohyoid, mylohyoid, and digastric; and the mucosal and submucosal surfaces at the base of the tongue. Neural coordination of these complex activities is mediated primarily by cranial nerves V, VII, IX, X, XI, and XII [14].
This detailed understanding helps refine radiotherapy planning, focusing on protecting these critical structures to reduce the risk of dysphagia while maintaining effective cancer treatment outcomes.
CT-Based Muscle Thickness Assessment
Further insights into the structural impact of radiotherapy are illuminated through serial CT studies. Post-treatment imaging has documented notable increases in the thickness of the pharyngeal constrictors and the laryngeal apparatus, indicative of radiation-induced edema and fibrosis [15]. Conversely, the suprahyoid muscles, the longitudinal pharyngeal muscles (up to their juncture with the pharyngeal constrictors), and the mucosal and submucosal layers of the tongue and its base have not demonstrated significant changes following radiotherapy.
This accumulated evidence from radiological and functional assessments underscores the importance of certain anatomical structures in the context of dysphagia. Specifically, the pharyngeal constrictors, the cricopharyngeus, the musculature at the esophageal inlet, and the glottic and supraglottic components of the larynx are identified as critical DARS. These structures require meticulous contouring during radiotherapy planning to minimize treatment-related morbidity while ensuring the effectiveness of oncological outcomes [16,17].
Dose-Dependent Dysphagia Risk
The clinical significance of DARS is underscored by a prospective study conducted by Mogadas et al., which assessed 54 patients with locally advanced oropharyngeal cancer undergoing IMRT [18]. This research highlighted a significant relationship between late dysphagia and the radiation dose administered to the lower pharyngeal constrictor.
Among the patients receiving a radiation dose of 55 Gy or higher, 14 (64%) experienced dysphagia of grade 2 or lower, while 8 (36%) developed more severe dysphagia of grade 3 or higher. Conversely, among those exposed to less than 55 Gy, 22 (69%) had dysphagia of grade 2 or lower, compared to 10 (31%) who suffered from grade 3 or higher dysphagia.
Furthermore, the dose delivered to the middle pharyngeal constrictor was a significant predictor of late dysphagia. Statistically significant correlations were observed at 6 months (p = 0.000), 12 months (p = 0.005), and 18 months (p = 0.034) post-treatment, highlighting the critical nature of radiation management in these regions. By 24 months, however, the association was no longer significant (p = 0.374), which may indicate compensatory neuromuscular adaptations or a delayed recovery process.
Interestingly, the upper pharyngeal constrictor demonstrated minimal relevance to dysphagia outcomes, suggesting a more limited role in post-radiotherapy swallowing dysfunction. This observation emphasizes the necessity of prioritizing dose reduction strategies for the middle and lower pharyngeal constrictors to mitigate long-term swallowing complications effectively.
Strategic Delineation of DARS in Radiotherapy
This detailed analysis underscores the necessity to accurately identify and safeguard anatomical structures vulnerable to radiation, crucial for maintaining swallowing functionality. Modern radiotherapy meticulously focuses on protecting DARS, balancing cancer control with minimizing treatment-related complications. Insights from videofluoroscopic and CT assessments guide the precise contouring and protection of vital structures like pharyngeal constrictors and laryngeal components. This targeted approach not only prepares the groundwork for the next discussion on the delineation of DARS in radiotherapy planning but is also essential for minimizing dysphagia risks and enhancing outcomes for head and neck cancer patients.
With a clear understanding of treatment-induced changes, attention must now turn to the accurate delineation of anatomical targets essential for preserving swallowing function. Effective radiotherapy planning requires precise identification and contouring of structures involved in deglutition and aspiration. This process should be conducted using CT simulation images before radiotherapy planning begins, enabling accurate assessment of radiation doses to these regions. In the context of Do-IMRT, meticulous delineation is critical to reduce the risk of geographic miss in the planning target volume and to balance oncologic efficacy with functional preservation.
Although more than 30 muscle pairs and six cranial nerves contribute to the intricate coordination of swallowing, not all require contouring for dosimetric analysis. Current consensus highlights a core set of anatomical structures that are most relevant to dysphagia and aspiration, including the pharyngeal constrictors, cricopharyngeus muscle, esophageal inlet musculature, cervical esophagus, and larynx (Table 1, Figure 1) [7].
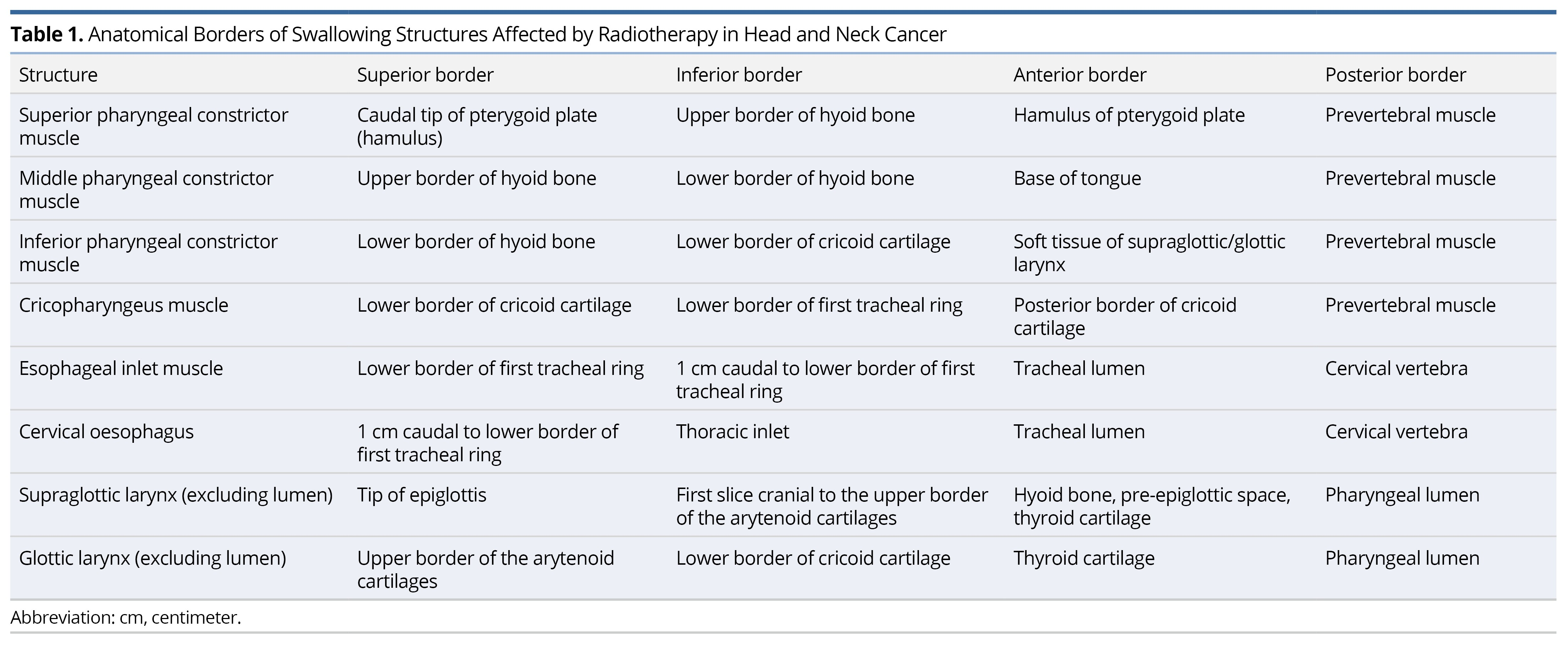
Pharyngeal Constrictors
The pharyngeal wall consists of two distinct muscle layers: an external circular layer composed of the pharyngeal constrictors and an internal longitudinal layer formed by the levator muscles. The pharyngeal constrictors are subdivided into three segments: the superior constrictor extends from the caudal aspect of the lateral pterygoid plate and pterygoid hamulus to the upper border of the hyoid bone; the middle constrictor spans the cranial to caudal margin of the hyoid bone; and the inferior constrictor originates at the lower hyoid margin and continues to the inferior border of the cricoid cartilage. Posteriorly, these muscles abut the prevertebral musculature, while anteriorly they border the pharyngeal lumen [19].
Cricopharyngeus
The cricopharyngeus muscle originates at the caudal edge of the cricoid cartilage and extends approximately 1 cm caudally to the level of the first tracheal ring. Its anterior margin attaches to the posterior outer surface of the cricoid cartilage, and its posterior extent is defined by the prevertebral musculature [20].
Esophageal Inlet Muscles
The esophageal inlet represents the proximal segment of the esophagus and is especially vulnerable to radiation-induced stricture. It begins at the inferior aspect of the cricopharyngeus muscle and extends approximately 1 cm caudally. The anterior margin is defined by the trachea and the posterior boundary by the prevertebral muscles.
Cervical Esophagus
The cervical esophagus begins 1 cm below the inferior margin of the cricoid cartilage and continues to the thoracic inlet. On axial CT, it is readily identifiable without the need for anterior and posterior margin delineation, simplifying its inclusion in treatment planning [21].
Larynx
The larynx is anatomically divided into supraglottic, glottic, and subglottic regions. The supraglottis includes the epiglottis, supraglottic adductor muscles, aryepiglottic folds, arytenoid cartilages, and false vocal cords. Delineation should encompass the epiglottis, median glossoepiglottic fold, supraglottic adductors, hyoepiglottic ligament, and aryepiglottic folds, with the cranial border set at the tip of the epiglottis and the caudal border at the upper edge of the arytenoid cartilages [22].
For the glottic larynx, the superior boundary aligns with the upper margin of the arytenoid cartilages. Included structures comprise the arytenoid cartilages, glottic adductor muscles, and both true and false vocal cords. The cricoid cartilage itself is excluded, except for the soft tissue between the thyroid and cricoid cartilages, which is included as part of the glottic larynx.
Optimizing DARS Preservation
This methodical approach to anatomical delineation does more than just identify critical areas; it establishes the essential structural framework required for optimal preservation of DARS and reduces radiation-induced swallowing dysfunction in head and neck cancer patients. By precisely defining these vital structures during the radiotherapy planning process, we effectively enhance the equilibrium between oncological efficacy and the preservation of key functional capacities. This strategy is instrumental in supporting improved patient outcomes, ensuring that treatment rigorously adheres to both therapeutic objectives and quality of life considerations.
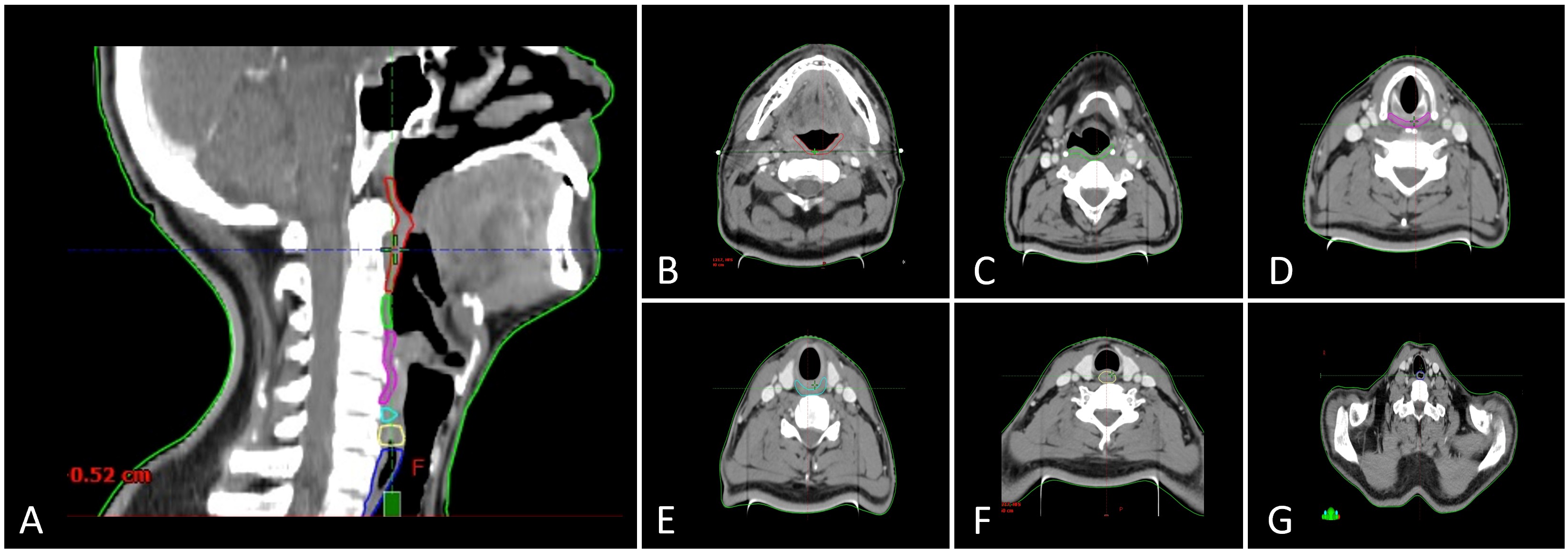
Figure 1. Anatomical delineation of swallowing-related organs at risk on computed tomography (CT). (A) Sagittal CT section with color-coded contours delineating key swallowing-related organs at risk: superior pharyngeal constrictor (red), middle pharyngeal constrictor (deep green), inferior pharyngeal constrictor (magenta), cricopharyngeus (cyan), esophageal inlet (yellow), and cervical esophagus (deep blue). (B) Axial CT section at the level of the superior pharyngeal constrictor (red). (C) Axial CT section showing the middle pharyngeal constrictor (deep green). (D) Axial CT section depicting the inferior pharyngeal constrictor (magenta). (E) Axial CT section highlighting the cricopharyngeus (cyan). (F) Axial CT section illustrating the esophageal inlet (yellow). (G) Axial CT section of the cervical esophagus (deep blue).
Having clarified the significance of DARS in radiation-induced dysphagia and addressed their precise delineation in radiotherapy planning, the discussion now advances to establishing appropriate dose constraints. More than 30 muscle pairs and six cranial nerves coordinate the swallowing process. However, current evidence indicates that radiation doses delivered to specific structures immediately beneath the mucosa and submucosa primarily cause acute and chronic dysphagia. Among these, the pharyngeal constrictors, glottis, and supraglottis are particularly vulnerable. Defining evidence-based dose thresholds for these critical structures is therefore essential. Implementing these constraints enables clinicians to optimize tumor control while effectively minimizing treatment-related swallowing dysfunction, thus enhancing patient quality of life.
Dose Constraints for the Pharyngeal Constrictors
To establish safe dose thresholds for critical structures, various clinical studies have explored the dose-volume effects on swallowing outcomes. Among these, Mazzola et al. [23] conducted a comprehensive analysis of the risk of dysphagia associated with radiation exposure to the constrictor muscles in 56 patients with head and neck cancer undergoing IMRT. This study pinpointed dose-volume parameters predictive of grade 2 or higher dysphagia, both acute and chronic, providing essential guidance for treatment planning. Notably, the study defines V50 as the volume percentage of a tissue or organ receiving at least 50 Gy of radiation, which is a critical parameter in assessing radiation exposure and predicting tissue response.
Effects on the superior constrictor muscle
No significant correlations were initially observed between dosimetry of the superior constrictor muscle and acute dysphagia. However, data collected three months post-radiotherapy revealed significant associations. Specifically, a maximum dose exceeding 50 Gy, a mean dose above 60 Gy, and a V50 greater than 70% were significantly linked to increased toxicity. By six months, only the V50 exceeding 70% maintained statistical significance, with no evident correlations at the 12-month assessment [23].
Effects on the middle constrictor muscle
In contrast, the middle constrictor muscle demonstrated a stronger association with early dysphagia. A maximum dose above 50 Gy, a mean dose exceeding 60 Gy, and a V50 greater than 70% were significantly related to grade 2 or higher dysphagia during radiotherapy and at three months post-treatment. These associations did not persist beyond the six-month mark, suggesting an acute effect [23].
Effects on the inferior constrictor muscle
For the inferior constrictor muscle, none of the evaluated dosimetric parameters were statistically significant in correlating with dysphagia at any assessed time points, indicating a relatively minor role of this segment in swallowing dysfunction under typical dose exposures [23].
Effects on the cricopharyngeus muscle
The cricopharyngeus muscle exhibited limited dosimetric sensitivity. Only a maximum dose exceeding 60 Gy was significantly associated with an increased risk of grade 2 or higher dysphagia during radiotherapy. No other dose-volume parameters demonstrated significant correlations with either acute or delayed swallowing impairment [23].
Dose Constraints for the Larynx
Laryngeal edema is a predominant factor in dysphagia following radiotherapy for head and neck cancer. Sanguineti et al. [24] recommended minimizing both the mean dose to the larynx and the V50, with suggested thresholds of less than 43.5 Gy and 27%, respectively.
Levendag et al. [20] found that a median laryngeal dose of 50 Gy was predictive of a 20% likelihood of dysphagia in patients treated with either three-dimensional conformal radiotherapy (3DCRT) or IMRT. Doornaert et al. [25] observed a steep dose-response relationship with the laryngeal wall beyond 45 Gy, suggesting that 45 Gy is the optimal threshold for minimizing the risk of swallowing difficulties.
Strategic Dose Management for Dysphagia Prevention
This discussion highlights the importance of defining precise dose constraints for key swallowing structures in head and neck cancer radiotherapy. By adhering to evidence-based thresholds, especially for the pharyngeal constrictors, glottis, and supraglottis, clinicians can effectively reduce the incidence of radiation-induced dysphagia. Implementing these strategic dose limitations is crucial for preserving essential functions and patient quality of life, thereby achieving a better balance between oncological efficacy and functional preservation.
Recent advancements in radiotherapy focus on enhancing the therapeutic ratio by reducing radiation-induced toxicity, without compromising oncological efficacy. Among these advancements, IMRT stands out as a pivotal development, particularly its dysphagia-optimized variant, Do-IMRT, which prioritizes the preservation of swallowing function. By enabling highly conformal dose delivery that selectively spares critical structures involved in deglutition, Do-IMRT overcomes significant limitations associated with earlier radiotherapy techniques.
With the increase in long-term survivorship in head and neck cancer, there is a growing recognition of the persistent functional impairments resulting from treatment. Late-onset dysphagia, in particular, garners attention due to its significant impact on patients’ quality of life. This concern spurs the development of Do-IMRT strategies aimed at minimizing radiation exposure to pharyngeal constrictors and other relevant anatomical structures [26]. Concurrently, technological innovations such as arc-based IMRT and adaptive radiotherapy open new paths for refining dose delivery, thereby enhancing the potential to alleviate treatment-related dysphagia. Subsequent sections delve into the clinical rationale, dosimetric innovations, and burgeoning evidence supporting the implementation of Do-IMRT.
Advantages of IMRT
Historically, the management of head and neck cancers often relied on conventional techniques such as the three-field approach, which includes bilateral parallel-opposed fields and a low anterior neck field, or the anterolateral wedge pair technique. These traditional methods, however, lacked the capability for selective dose modulation to anatomical structures involved in swallowing, leading to increased rates of treatment-associated toxicities. The advent of IMRT has effectively addressed these limitations, significantly enhancing both clinical outcomes and patient quality of life.
IMRT represents an advanced evolution of 3DCRT, offering particular benefits for treating tumors with concave or complex geometries adjacent to radiosensitive tissues. The core advantages of IMRT stem from its ability to emit radiation beams of non-uniform intensities, facilitated by computerized inverse planning. Unlike traditional radiotherapy, which utilizes uniformly intense radiation beams, IMRT divides each beam into numerous smaller beamlets. The intensity of each beamlet is independently adjustable, permitting highly conformal dose distributions. This capability enables precise dose shaping around irregular tumor volumes and markedly reduces radiation exposure to surrounding normal structures.
Transatlantic Prospective Trial: IMRT and Dysphagia
A prospective trial was conducted at two prominent transatlantic locations: the University of Michigan and Erasmus University in Rotterdam [27]. At the University of Michigan, 36 patients with predominantly oropharyngeal tumors underwent IMRT, which effectively spared the uninvolved pharyngeal constrictors, glottis, and supraglottis while ensuring comprehensive target coverage. Conversely, at Erasmus University, 77 patients received various radiotherapeutic interventions, including brachytherapy which was administered to 52% of these patients.
Dose-volume effects on dysphagia
In the Michigan cohort, all patients who exhibited aspiration received a mean pharyngeal constrictor dose exceeding 60 Gy. Critical volumetric indicators included V65, the percentage of volume receiving 65 Gy or more, which surpassed 50% for the pharyngeal constrictors; and V50, the percentage of volume receiving 50 Gy or more, also exceeding 50% for the larynx. Notably, the superior constrictor was most strongly correlated with both observer-rated and patient-reported dysphagia scores. In the Rotterdam cohort, univariate analysis identified a significant correlation between dysphagia and the doses delivered to the superior and middle pharyngeal constrictors. Further, multivariate analysis confirmed brachytherapy as the primary modality associated with a reduction in symptoms, significantly lowering the radiation doses to the superior and middle constrictors.
Long-Term Dysphagia Outcomes in IMRT
A separate prospective trial conducted by Feng and colleagues at the University of Michigan [28] assessed the efficacy of IMRT in terms of survival and its capacity to preserve swallowing function by sparing critical anatomical structures. Seventy-three patients with stage III to IV oropharyngeal cancer underwent chemoradiotherapy using IMRT, specifically tailored to minimize radiation exposure to the pharyngeal constrictors and larynx. The study reported three-year disease-free survival and loco-regional recurrence-free survival rates of 88% and 96%, respectively.
Functional swallowing assessments post-IMRT
Initial assessments revealed a significant worsening in both observer-rated and patient-reported dysphagia scores during the early post-therapy period. However, these scores gradually improved over 12 months, stabilizing in the second year. By the 12-month evaluation, nearly all observer-rated scores were 0 or 1, with a score of 1 indicating mild dysphagia that did not necessitate dietary modifications. In contrast, video-fluoroscopy assessments indicated a progression from mild dysfunction before therapy to mild-to-moderate dysfunction immediately post-therapy, with no subsequent recovery observed. This decline was partly attributed to aspiration events, which occurred shortly after therapy commencement and persisted for two years. Notably, most aspirations were silent, not triggering a cough reflex and thus remained undetected by the patients. The observed correlation between aspiration rates and radiation doses to swallowing structures indicates that further dose reductions could potentially mitigate dysphagia. Such reductions might be achievable through refined IMRT planning techniques, including split-field IMRT [29].
Do-IMRT: Concept and Implementation
While IMRT has significantly enhanced dose conformality and reduced toxicity to adjacent normal tissues, further refinements have been directed towards selectively sparing swallowing-related structures. This focus has culminated in the development of Do-IMRT, which incorporates anatomical specificity into the planning process to further minimize functional impairment. In this technique, areas of the pharyngeal constrictors and larynx outside the high-dose target volume are contoured separately. Specific dose constraints are then applied to these structures during inverse planning optimization. The primary objective of Do-IMRT is to minimize radiation-induced damage to unaffected swallowing structures while ensuring adequate dose delivery to the target volume [30].
Do-IMRT Enhances Dosimetric Precision
A pivotal study by Eisbruch et al. [7] evaluated radiotherapy plans for 20 patients with advanced stage III to IV head and neck cancer undergoing concurrent chemoradiotherapy using three different techniques: 3DCRT, standard IMRT, and Do-IMRT. Uniform dose specifications and constraints were maintained across all techniques, except in the case of Do-IMRT, where an additional cost function was integrated to reduce radiation exposure to the pharyngeal constrictors and larynx without compromising the dose to the target volume.
The analysis indicated that IMRT reduced the V50 of the pharyngeal constrictors by 10% compared to 3DCRT. Do-IMRT achieved an additional 10% reduction in this parameter. Similarly, the V50 for the larynx was decreased by 7% with IMRT in comparison to 3DCRT, and Do-IMRT further lowered this volume by an additional 11%. These results underscore the dosimetric benefits of Do-IMRT in preserving key swallowing structures while maintaining effective tumor control.
The DARS Trial: First RCT on Do-IMRT
The DARS trial (CRUK/14/014) [31], a phase 3 multicenter randomized controlled trial, compared Do-IMRT with standard IMRT in patients diagnosed with stage I-IVB oropharyngeal or hypopharyngeal cancer undergoing chemoradiotherapy. This inaugural RCT focused on swallow-sparing radiotherapy aimed to enhance long-term swallowing function by specifically delineating pharyngeal constrictor muscles as organs at risk in Do-IMRT and optimizing treatment plans to adhere to strict dose constraints. Both treatment arms received 65 Gy delivered in 30 fractions.
At the 12-month mark, Do-IMRT demonstrated superior patient-reported swallowing outcomes, evidenced by higher MD Anderson Dysphagia Inventory (MDADI) composite scores (77.7 vs. 70.6; mean difference 7.2; p < 0.037). This advantage, attributed to reduced radiation doses to the inferior pharyngeal constrictors (28.4 Gy vs. 49.8 Gy) and to the superior and middle pharyngeal constrictors (49.7 Gy vs. 57.2 Gy; both p < 0.0001), was evident by 6 months and sustained through 24 months (adjusted p = 0.0030 at 12 months). These outcomes were assessed using MDADI from 3 to 24 months and videofluoroscopy at 12 and 24 months.
This groundbreaking trial positions Do-IMRT as a potential standard in radiotherapy, significantly enhancing the quality of life for survivors. However, the focus on HPV-positive oropharyngeal cases and the 24-month follow-up period limit the scope for broader and long-term insights. Future research should aim to extend the follow-up duration, diversify patient cohorts, and explore the integration of proton therapy to advance precision in radiotherapy for functional preservation.
Multimodal Strategies to Reduce Dysphagia
Despite advancements in Do-IMRT, technological enhancements alone prove insufficient to completely eradicate dysphagia. Complementary strategies are crucial and involve personalized adjustments to treatment intensity informed by predictors of tumor control, enhanced cytoprotective strategies, targeted radiosensitization, and the timely initiation of swallowing therapy as clinically indicated. These multimodal interventions have the potential to significantly reduce both the severity and prevalence of treatment-related dysphagia.
While Do-IMRT marks a significant advance in reducing treatment-related dysphagia through precise dose modulation and anatomical sparing, technological refinements alone prove insufficient to completely eradicate late toxicities. Among patients with low-risk head and neck cancer, although long-term disease control often remains excellent, functional morbidity continues to be a significant issue. Notably, the five-year locoregional control rate following surgery exceeds 90%; however, about 30% of these patients still experience grade III chronic toxicities, including dysphagia, xerostomia, and trismus, which severely impact quality of life [32].
In response to these challenges, de-intensification strategies have been suggested for this subgroup with a favorable risk profile. These strategies focus on reducing the radiation dose or the treatment volume to mitigate adverse effects without sacrificing oncological efficacy. The subsequent section delves into pivotal clinical trials that explore the feasibility, dosimetric impact, and potential trade-offs associated with radiotherapy de-intensification in the postoperative scenario.
DIREKHT Trial: Reduced Dose and Volume
In the multi-institutional DIREKHT Trial, Haderlein et al. [33] tested the efficacy of reducing both the radiation dose and treatment volume in a highly selective group of head and neck cancer patients. These patients were chosen for their low-risk tumor characteristics: primary tumors classified as pT2 or smaller, resection margins exceeding 5 mm, and no evidence of lymphovascular or perineural invasion. The radiotherapy regimen for this subgroup was confined to 56 Gy to the primary tumor bed, with contralateral neck irradiation explicitly excluded for patients who had fewer than three ipsilateral lymph node metastases and no clinically evident contralateral nodal involvement.
The study’s findings, based on an analysis of thirty patients, indicated significant reductions in radiation exposure. Notably, the middle and inferior pharyngeal constrictors, cricopharyngeal muscles, glottis, supraglottic regions, and contralateral salivary glands received over 10 Gy less radiation than in standard protocols (p < 0.000). The volume of elective neck nodal irradiation was also significantly decreased, measuring only 662 mL compared to 1166 mL in standard treatment plans.
These results underscore the potential of de-intensified treatment protocols to significantly reduce the risk of chronic toxicities such as dysphagia, xerostomia, and trismus without compromising cancer control effectiveness. By limiting radiation exposure to non-targeted areas, this approach promises to enhance the quality of life for patients with favorable prognostic indicators, suggesting a pivotal shift in treatment strategies for low-risk head and neck cancer patients.
CheckRad-CD8 Trial: Modified Contour Approach
Weissmann et al. [34] conducted an analysis on the first 30 patients in the CheckRad-CD8 trial at a single center, implementing a novel contouring technique in radiation therapy. Unlike traditional methods, this trial maintained the total radiation dose while modifying the contouring of the low-dose (56 Gy) volume to focus exclusively on the elective nodal levels adjacent to the involved lymph nodes. This departure from the conventional elective nodal volume, which typically encompasses broader regions based on N-stage, aimed to concentrate the dose more precisely and reduce unnecessary radiation exposure to adjacent structures.
Dose-volume calculations indicated a significant reduction in radiation volume: the median elective treatment area receiving 56 Gy was reduced from 1091.9 cm³ to 750.3 cm³ (p < 0.001), and the high-risk area receiving 63 Gy decreased from 754.3 cm³ to 368.77 cm³ (p < 0.001). Most notably, these modifications resulted in significantly lower radiation exposure to the middle and inferior pharyngeal constrictors, the cricopharyngeus muscle, and the glottic and supraglottic larynx.
These findings suggest that the modified contouring approach is designed to conserve therapeutic efficacy while substantially diminishing the risk of radiation-induced damage and associated toxicities in non-target tissues. By reducing unnecessary radiation exposure, this technique could potentially enhance patient outcomes by decreasing the incidence of treatment-related complications such as dysphagia and xerostomia, and possibly trismus, thus improving overall quality of life for patients undergoing radiotherapy for head and neck cancers.
Future Considerations in Radiotherapy De-intensification
The primary objective of radiotherapy de-intensification trials in head and neck cancer is to evaluate the feasibility and extent of reducing radiation doses or treatment volumes, particularly for anatomical structures associated with dysphagia. However, current trials, including DIREKHT and CheckRad-CD8, have not yet provided definitive data on locoregional failure rates resulting from these reductions compared with standard treatment protocols. Instead, available reports have focused primarily on preliminary dosimetric and volumetric outcomes. Most trials remain in active accrual phases, and the published data thus far provide initial insights into reductions in radiation exposure rather than conclusive evidence regarding oncological endpoints.
Further follow-up and larger patient cohorts are necessary to determine whether reduced-dose or reduced-volume adjuvant radiotherapy can achieve comparable locoregional control in low-risk postoperative head and neck cancer patients. Ultimately, future studies should confirm whether radiotherapy de-intensification strategies effectively reduce chronic toxicities, such as dysphagia, xerostomia, and trismus, without compromising clinical efficacy and patient survival.
De-intensification strategies, including dose and volume reduction approaches exemplified by the DIREKHT [33] and CheckRad-CD8 [34] trials, have shown promising preliminary results. However, their effectiveness in mitigating radiation-induced dysphagia while preserving oncologic outcomes remains uncertain. This uncertainty necessitates the exploration of alternative advanced radiotherapy modalities. Proton therapy, distinguished by its unique physical properties and advantageous dose distribution, presents itself as a potential solution meriting thorough investigation. Subsequent sections assess whether proton therapy can effectively address these unresolved challenges and ameliorate treatment-related dysphagia in patients with head and neck cancer.
Proton Therapy Overview
Protons are positively charged particles distinguished by their unique capability to deposit maximum energy at a specific tissue depth, reaching a peak at what is known as the Bragg peak, followed by a rapid dose decline. To effectively cover tumors at various depths, proton therapy employs energy modulation to create an extended Bragg peak, ensuring comprehensive coverage of the entire target volume. Intensity-modulated proton therapy (IMPT) further refines this technique by adjusting the intensity of individual proton beamlets, facilitating highly conformal dose distributions that enhance the protection of surrounding healthy tissues. Although proton therapy optimizes target coverage and minimizes radiation exposure to adjacent organs at risk, it may also elevate the entrance dose and the risk of radiation-induced dermatitis, a consequence of the beam modulation requirements.
Challenges in Proton Therapy
A significant limitation of proton therapy in the treatment of head and neck cancer is the scarcity of robust clinical data, primarily attributable to challenges like setup variability and anatomical uncertainties. Consequently, there is a lack of Level I evidence to support its widespread adoption in clinical practice [35]. Existing clinical evidence is predominantly derived from patient populations with non-squamous cell histologies or those undergoing re-irradiation, under conditions that inherently differ from those in primary squamous cell carcinoma scenarios [36].
Despite these limitations, proton therapy offers substantial theoretical advantages in managing specific tumor sites, notably tumors at the skull base, as well as nasal, paranasal sinus, and oropharyngeal squamous cell carcinomas. The potential benefits of proton therapy in these cases stem from the anatomical complexity and proximity of the tumors to critical structures such as the brainstem, spinal cord, optic nerves and chiasm, and temporal lobes [37–39]. Careful patient selection and further prospective trials are essential to definitively ascertain the clinical scenarios in which proton therapy provides significant advantages over current photon-based therapies.
Prospective Evaluation of IMPT in Oropharyngeal Cancer
Gunn and colleagues conducted a single-institution prospective study at MD Anderson Cancer Center to evaluate the oncologic, toxicity, and functional outcomes in patients with oropharyngeal squamous cell carcinoma treated using IMPT between March 2011 and July 2014 [40]. The cohort included 50 patients, of whom 44 (88%) had confirmed HPV-positive tumors; HPV status was undocumented in five patients. Most patients presented with advanced-stage disease, with 98% classified as stage III or IV. At a median follow-up of 29 months, the two-year overall survival and progression-free survival rates were 94.5% and 88.6%, respectively, reflecting favorable oncologic efficacy.
IMPT demonstrated an encouraging toxicity profile, with no grade 4 or 5 adverse events observed. The most frequently reported acute grade 3 toxicities included oral mucositis (58%, 29 patients), radiation dermatitis (46%, 23 patients), and dysphagia (24%, 12 patients). During treatment, 11 patients (22%) required gastrostomy tube placement; notably, none remained dependent on enteral feeding at last follow-up. Late grade 3 toxicities comprised dysphagia in six patients (12%), persistent xerostomia in one patient (2%), and oral mucositis in one patient (2%). Median weight loss during treatment was 7.4%.
These findings reinforce the potential of IMPT to achieve robust disease control while minimizing severe treatment-related toxicities in patients with HPV-positive oropharyngeal carcinoma. The observed reduction in long-term gastrostomy dependence and swallowing dysfunction indicates meaningful quality-of-life benefits, strongly supporting further investigation of proton therapy in this clinical context.
IMPT vs. IMRT: Comparative Analysis
As the potential of IMPT to manage disease effectively while minimizing adverse effects becomes apparent, a detailed comparative analysis between IMPT and more traditional IMRT is crucial to elucidate distinctions in efficacy and toxicity profiles. Blanchard et al. conducted a case-matched comparative study evaluating the clinical outcomes of 50 patients with oropharyngeal cancer treated with IMPT versus 100 matched patients treated with IMRT [41]. The results demonstrated a lower incidence of severe weight loss and gastrostomy tube dependence in the IMPT cohort. At three years, the IMPT group achieved favorable oncologic outcomes, with overall survival and progression-free survival rates of 94.3% and 85.8%, respectively. In terms of treatment-related toxicity, 12 IMPT patients required gastrostomy tubes, and 39 experienced grade 2–3 fatigue during therapy. At three months post-treatment, four patients had lost more than 20% of their baseline body weight, and 21 reported grade 2–3 xerostomia.
These results indicate that IMPT may provide significant advantages over IMRT in reducing treatment-related morbidity without compromising oncologic efficacy in selected patients with oropharyngeal cancer. While these data are promising, the evidence base is still limited, especially concerning long-term functional outcomes and disease control across broader patient populations. Additionally, the applicability of IMPT beyond HPV-positive oropharyngeal cancer raises questions. Current evidence supporting its use in treating laryngeal and hypopharyngeal malignancies is minimal and is confounded by unique anatomical, biological, and technical challenges associated with these tumor sites.
To elucidate the role of proton therapy in head and neck oncology, further prospective randomized trials are indispensable. Such studies should compare IMPT and IMRT across a broader spectrum of disease subsites and clinical stages. They must also integrate patient-reported outcomes, cost-effectiveness analyses, and long-term toxicity endpoints to comprehensively assess the clinical utility of IMPT. Expanding the evidence base is crucial to establish precise indications for proton therapy and to optimize treatment strategies that prioritize both tumor control and patient quality of life.
Potential of Proton Therapy to Reduce Dysphagia
Emerging evidence indicates that proton therapy, particularly IMPT, may reduce dysphagia in selected patients with head and neck cancer. Studies in HPV-positive oropharyngeal cancer report oncologic outcomes comparable to intensity-modulated radiation therapy (IMRT), but with lower rates of swallowing dysfunction, weight loss, and gastrostomy tube dependence. These advantages stem primarily from the superior dose conformity of proton therapy, better sparing critical swallowing structures such as the pharyngeal constrictors and larynx.
However, current evidence largely arises from retrospective studies, often limited by small sample sizes and insufficient long-term follow-up. The effectiveness of proton therapy in other head and neck subsites, such as the larynx and hypopharynx, also remains inadequately studied. High-quality prospective trials focusing on functional endpoints are needed to clearly define its role. Until stronger evidence is available, proton therapy should be viewed as a promising but not yet fully validated approach for dysphagia mitigation in head and neck cancer.
Dysphagia is increasingly recognized as a critical factor influencing long-term quality of life in survivors of head and neck cancer, particularly in the context of organ preservation treatment approaches. As therapeutic strategies evolve to reduce morbidity, accurate assessment and management of swallowing impairment have become essential. Effective dysphagia evaluation informs clinical decisions, guides rehabilitation, and addresses patient concerns, especially those undergoing postoperative radiotherapy who often fear lasting swallowing dysfunction. A thorough and structured assessment, incorporating both patient-reported outcomes and objective clinical measures, is therefore crucial. This section reviews current methods for evaluating dysphagia, highlighting validated tools that facilitate individualized interventions and enhance patient outcomes.
Subjective Dysphagia Assessment Methods
A thorough evaluation of dysphagia necessitates the integration of subjective patient experiences with clinically observable indicators. Patient-reported outcome measures constitute an essential component of this assessment, capturing the direct impact of swallowing dysfunction on patients’ quality of life and daily activities. Subjective evaluations not only provide valuable insights into symptom severity but also inform the necessity for further diagnostic procedures and therapeutic interventions. The following discussion outlines validated instruments commonly utilized to assess dysphagia from the patient’s perspective, detailing their structure, scoring systems, and clinical applicability within head and neck oncology care.
MDADI: patient-reported dysphagia assessment
The MDADI, originally developed as the MD Anderson Dysphagia Inventory, is the most widely employed patient-reported outcome measure for evaluating dysphagia and its impact on quality of life in patients following radiotherapy. The instrument consists of 20 items divided into four domains: global, emotional, functional, and physical. The global domain includes a single item assessing the overall impact of swallowing difficulties on the patient’s quality of life. Patients rate each item on a five-point Likert scale ranging from 1 (“strongly agree,” indicating significant impairment) to 5 (“strongly disagree,” indicating minimal impairment). The global subscale (the first item) is scored independently, while the remaining 19 items are summed and averaged. This average score is multiplied by 20 to yield a total score ranging from 0 (severe swallowing impairment) to 100 (optimal swallowing function), with higher scores representing better swallowing-related quality of life [42].
EORTC QLQ-H&N35: cancer-specific quality of life evaluation
The European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-Head and Neck 35 (EORTC QLQ-H&N35) is a validated, disease-specific extension of the general EORTC QLQ-C30 quality of life instrument, tailored explicitly for head and neck cancer patients. The questionnaire comprises 35 items administered prior to treatment initiation and subsequently at regular intervals for up to 3.5 years after treatment [43]. The initial 30 items employ a four-point Likert scale (“not at all,” “a little,” “quite a bit,” and “very much”), while the final five items use a binary (“yes/no”) format. Higher total scores reflect greater symptom severity or functional impairment. The instrument has been validated in a multicenter cohort of 500 newly diagnosed patients with head and neck cancer in Norway, Sweden, and the Netherlands, demonstrating robust compliance and sensitivity to changes in symptomatology over time [43].
FACT-H&N: comprehensive quality of life measurement
The Functional Assessment of Cancer Therapy-Head and Neck (FACT-H&N, version 4) is a multidimensional, patient-reported quality of life instrument specifically developed for patients with head and neck malignancies. The FACT-H&N consists of 27 core items (FACT-General, FACT-G) assessing four domains: physical, social/family, emotional, and functional well-being. Additionally, it includes 12 head and neck-specific items designed to evaluate symptoms associated with disease and treatment. Responses are recorded on a five-point Likert scale (0–4). Domain-specific subscale scores are calculated individually, and an aggregate global quality of life score is generated by summing all responses. Higher scores represent better overall quality of life [44–46].
Objective Dysphagia Assessment Methods
Subjective tools provide valuable insights into patients’ perceptions of dysphagia and its effects on quality of life. However, these tools offer limited information regarding the physiological mechanisms underlying swallowing dysfunction. Consequently, objective assessment methods are critical for accurately characterizing swallowing biomechanics, detecting silent aspiration, and evaluating treatment-related changes over time. These techniques allow clinicians to directly observe swallowing dynamics and quantify impairment with high precision, thereby informing clinical decision-making and guiding tailored intervention strategies. This section outlines the principal objective modalities employed in clinical practice, emphasizing videofluoroscopic evaluation and validated scoring systems.
Videofluoroscopy for dysphagia evaluation
Among the most reliable methods for assessing dysphagia are the modified barium swallow study and videofluoroscopy. This real-time fluoroscopic technique evaluates swallowing function by analyzing bolus transit across a range of textures and consistencies, including thin-liquid barium and barium-coated solids. The procedure examines swallowing dynamics at baseline (pre-radiotherapy) and during subsequent follow-up assessments, focusing on the oral, pharyngeal, and upper esophageal phases.
Videofluoroscopy provides a comprehensive view of swallowing biomechanics, enabling the identification of specific impairments. The technique’s ability to capture dynamic imaging ensures precise evaluation of bolus flow and structural movement, making it a cornerstone of dysphagia assessment in clinical settings.
Penetration-aspiration scale scoring
Swallowing function is quantitatively assessed using the penetration-aspiration scale (PAS), an eight-point ordinal scale that grades the depth of bolus penetration beyond the vocal cords and the patient’s ability to clear it [47]. In head and neck cancer patients with moderate to severe dysphagia post-radiotherapy, videofluoroscopy commonly reveals impaired swallowing mechanics, including reduced posterior inversion of the tongue base, delayed laryngeal closure, diminished epiglottic inversion, decreased hyoid elevation, and weakened pharyngeal peristalsis.
A study by Agarwal et al. [48] investigated swallowing function in 47 patients with locally advanced head and neck cancer treated with definitive chemoradiotherapy. Of the 34 patients with low PAS scores (≤2) at baseline, 53% and 46% exhibited further deterioration at the two- and six-month follow-up assessments, respectively. Pre-treatment factors significantly associated with worse PAS scores included poor subjective swallowing function (p = 0.004), hypopharyngeal primary tumors (p = 0.05), and large tumor volume (p = 0.05). These findings underscore the utility of the PAS in tracking longitudinal changes in swallowing function.
Aspiration as a dysphagia assessment criterion
Aspiration, a key indicator of swallowing dysfunction, is often evaluated using videofluoroscopy. Eisbruch et al. [49] examined swallowing function in 29 patients with advanced head and neck cancer treated with high-dose radiotherapy (70 Gy in 35 fractions) and concurrent gemcitabine. Post-treatment assessments revealed that aspirations were predominantly “silent,” failing to trigger a cough reflex or eliciting a delayed and ineffective response. Aspiration was observed in 3 patients (14%) pre-treatment, increasing to 13 patients (65%) in early post-treatment assessments and persisting in 8 patients (62%) in late post-treatment assessments (p = 0.0002 for post-treatment vs. pre-treatment). This significant increase highlights the importance of objective measures in detecting clinically relevant changes that subjective reports may overlook.
Integrating Assessment Tools
This section examines dysphagia assessment in head and neck cancer survivors, combining subjective tools (MDADI, EORTC QLQ-H&N35, and FACT-H&N) with objective methods (videofluoroscopy and PAS). Subjective measures reveal patient-reported quality-of-life impacts, while objective approaches quantify swallowing biomechanics and identify silent aspiration. Studies highlight post-treatment deterioration and increased aspiration risk, emphasizing the need for both approaches to improve diagnostic accuracy and tailor interventions, enhancing patient outcomes.
Detailed assessments are crucial for understanding the anatomical and functional deficits contributing to dysphagia post-radiotherapy. However, diagnosis alone is insufficient to address the significant swallowing impairments many patients endure. Dysphagia rehabilitation is a vital yet often overlooked aspect of survivorship care, especially in developing countries where resources and specialized expertise are scarce. Effective management necessitates a multidisciplinary approach, involving rehabilitation therapists, oncologists, surgeons, prosthodontists, nurses, and social workers. Structured rehabilitation plans that include preventive strategies, early therapeutic interventions, and sustained maintenance are essential to restore swallowing function and enhance overall quality of life. This section elaborates on evidence-based rehabilitation strategies aimed at meeting these needs and fostering optimal functional recovery in patients with head and neck cancer.
Timing of Rehabilitation Exercises
The onset of fibrosis in irradiated structures is a leading cause of dysphagia, highlighting the need for early intervention through exercises that aim to minimize fibrosis. It is crucial for patients to start these exercises prior to beginning radiotherapy and to maintain them consistently throughout the treatment regime. Adherence on a daily basis is critical; typically, mucositis and generalized lethargy develop within two weeks of commencing radiotherapy, exacerbating dysphagia. Importantly, patients with a gastrostomy tube should persist with swallowing exercises to avert chronic dysphagia. Research indicates that patients who commence swallowing exercises pre- and during radiotherapy report a significantly enhanced quality of life compared to those who start post-treatment [50]. Following the completion of treatment, dysphagia rehabilitation therapists are tasked with conducting evaluations to identify the most beneficial maneuvers for each patient. To counteract the persistent effects of radiation-induced fibrosis, these exercises should be continued indefinitely.
Assessment Prior to Rehabilitation
Effective rehabilitation of dysphagia necessitates a comprehensive assessment of its underlying etiology and clinical manifestations to inform tailored treatment strategies. This diagnostic process employs targeted evaluations to systematically characterize swallowing function, establishing a critical foundation for subsequent therapeutic interventions.
Imaging modalities are pivotal in elucidating the causes of dysphagia in patients following radiotherapy. The two predominant techniques are the modified barium swallow study with videofluoroscopy and the fiberoptic endoscopic evaluation of swallowing. In the modified barium swallow study, patients ingest radiopaque boluses of varying consistencies, allowing clinicians to assess the oral, oropharyngeal, and pharyngeal phases of swallowing in real time. By contrast, the fiberoptic endoscopic evaluation of swallowing involves transnasal insertion of an endoscopic probe to provide detailed visualization of pharyngeal dynamics and vocal fold closure during deglutition. Of these, the modified barium swallow study is widely regarded as the gold standard for both diagnosis and management of dysphagia in post-radiotherapy patients due to its comprehensive assessment capabilities [51].
Postural Adjustments for Swallowing
Postural maneuvers constitute a key therapeutic approach to enhance bolus transit and mitigate aspiration risk in patients with dysphagia. Evidence suggests that such adjustments can prevent aspiration in approximately 81% of head and neck cancer patients following surgery or radiotherapy [52]. The choice of maneuver depends on the specific swallowing impairment and associated physiological deficits. Below, commonly utilized techniques are outlined, each designed to optimize bolus flow and bolster airway protection in individuals with swallowing dysfunction.
Chin-down maneuver
The chin-down maneuver involves lowering the chin toward the neck, modifying pharyngeal anatomy by displacing the anterior pharyngeal wall posteriorly and narrowing the airway entrance. This adjustment enhances epiglottic deflection and laryngeal vestibule closure, thereby reducing aspiration risk during the pharyngeal phase. It is particularly effective for patients exhibiting delayed pharyngeal swallow initiation or impaired airway protection.
Head-back maneuver
The head-back maneuver utilizes gravitational assistance to facilitate bolus propulsion from the oral cavity to the pharynx. Patients extend the head posteriorly, promoting posterior bolus movement and compensating for diminished lingual strength or impaired anterior-posterior tongue motion. This technique proves beneficial for individuals with oral phase deficits, particularly those struggling to initiate swallowing due to inadequate tongue control.
Head-rotation maneuver
In cases of unilateral vocal fold paralysis or asymmetric pharyngeal dysfunction, the head-rotation maneuver requires patients to turn their head toward the weaker side. This action compresses the impaired pharyngeal musculature, elevating intraluminal pressure and redirecting the bolus toward the more competent side. By minimizing the use of the dysfunctional pathway, this maneuver enhances pharyngeal clearance, reduces residual bolus accumulation, and strengthens airway protection.
Head-tilt maneuver
Distinct from head rotation, the head-tilt maneuver involves tilting the head toward the stronger side to leverage gravity in guiding the bolus along the more functional swallowing pathway. This approach improves oral and pharyngeal transit efficiency and is especially advantageous for patients with unilateral deficits in tongue control, pharyngeal peristalsis, or bolus manipulation. By optimizing clearance, it effectively minimizes aspiration risk.
Swallowing Maneuvers for Voluntary Control
Swallowing maneuvers enable patients to exert voluntary control over the oropharyngeal phase of swallowing, optimizing bolus clearance and airway protection. Videofluoroscopy is recommended to confirm the proper execution and efficacy of these maneuvers in individual patients, ensuring both safety and therapeutic benefit.
Supraglottic swallow maneuver
The supraglottic swallow maneuver involves a deep inhalation followed by breath-holding during swallowing, with an immediate forceful cough post-swallow to clear pharyngeal residue. This technique promotes early vocal cord closure prior to deglutition, reducing the risk of penetration or aspiration into the airway. It is particularly effective for patients with impaired laryngeal closure or delayed swallow initiation [53].
Mendelsohn maneuver
The Mendelsohn maneuver enhances laryngeal elevation and extends the duration of cricopharyngeal opening during swallowing. Patients initiate a swallow, sense the larynx’s upward movement, and hold it at its maximal elevation for approximately 3 seconds before completing the swallow. This technique is initially practiced without food to master execution, then integrated with oral intake. Studies demonstrate increased tongue base pressure, improved pharyngeal clearance, and reduced post-swallow residue in patients treated for head and neck cancer [54].
Tongue-hold maneuver
The tongue-hold maneuver, also termed the Masako maneuver, benefits patients post-radiotherapy for oropharyngeal cancer. Patients protrude the tongue, securing it gently between the central incisors, and swallow. This increases pressure at the tongue base and posterior pharyngeal wall, strengthening pharyngeal musculature. Due to potential increases in pharyngeal residue, it is typically recommended as a dry exercise without food to minimize aspiration risk [54].
Active Therapeutic Exercises for Swallowing Function
While postural and swallowing maneuvers mitigate aspiration risk and enhance oral intake, active therapeutic exercises target underlying physiological deficits to improve swallowing function, particularly in patients treated for head and neck cancer.
Oral tongue range-of-motion exercises
Tongue mobility and strength are essential for bolus control and propulsion. Patients extend the tongue maximally forward, holding for 2 seconds before relaxing. Additional movements include retraction, lateralization, and elevation, each sustained for 2 seconds. These exercises, repeated 5–10 times per session and performed 3–4 times daily, optimize tongue function [55].
Laryngeal range-of-motion exercises
Reduced laryngeal elevation significantly contributes to post-radiotherapy dysphagia. Exercises strengthening laryngeal musculature enhance airway protection and upper esophageal sphincter opening. One effective method involves falsetto phonation: patients slide up the pitch scale to their highest note, holding it for several seconds. This technique improves laryngeal excursion and reduces dysphagia-related complications.
Shaker maneuver
The Shaker maneuver strengthens suprahyoid muscles to enhance upper esophageal sphincter function and laryngeal elevation. In a supine position, patients perform three 1-minute sustained head raises, each followed by a 1-minute rest, then complete 30 repetitive head raises. This regimen improves sphincter opening, facilitating bolus transit and reducing post-swallow residue [56].
Assisted Rehabilitation Techniques
Unlike active exercises requiring voluntary effort, assisted rehabilitation techniques employ external stimulation to enhance neuromuscular activation and sensory input, supporting swallowing function. These approaches complement conventional exercises by targeting neuromuscular and sensory pathways, with neuromuscular electrical stimulation and thermal-tactile stimulation among the most studied modalities.
Neuromuscular electrical stimulation
Neuromuscular electrical stimulation employs surface electrodes placed on the neck to deliver electrical impulses, stimulating the hyoid and laryngeal muscles to enhance their elevation during swallowing. This technique has been evaluated to improve swallowing function in patients with dysphagia following radiotherapy. However, its efficacy remains uncertain. Although some studies report potential benefits in swallow mechanics, others observe no significant improvements in function or quality of life. Further research is required to identify optimal patient populations, refine stimulation parameters, and evaluate long-term outcomes in dysphagia rehabilitation [57].
Thermal-tactile stimulation
Thermal-tactile stimulation is employed to enhance the sensory trigger of the swallowing reflex. A laryngeal mirror, chilled in ice water for 10 seconds, is applied in vertical strokes to the anterior faucial arches bilaterally for five repetitions. This cold stimulus is intended to increase pharyngeal sensitivity and hasten the initiation of swallowing. Some studies suggest improvements in swallowing physiology among patients with head and neck cancer following radiotherapy, yet its clinical efficacy remains uncertain. Further well-controlled trials are required to determine its therapeutic role in dysphagia management [58].
Future Directions in Dysphagia Rehabilitation
The complexity of dysphagia following radiotherapy necessitates a multimodal rehabilitation strategy. Combining postural adjustments, swallowing maneuvers, active therapeutic exercises, and assisted techniques enhances swallowing function and minimizes aspiration risk. Protocols tailored to individual physiological impairments are essential for achieving sustained functional improvements. Future research should focus on optimizing these interventions, evaluating their long-term effectiveness, and investigating innovative approaches to improve quality of life for survivors of head and neck cancer.
This narrative review synthesizes existing literature on radiation-induced dysphagia, yet its design presents several inherent limitations. First, the narrative approach integrates studies with heterogeneous methodologies without systematic quantitative analysis, which may introduce selection bias and limit reproducibility. Second, variability in patient populations, tumor characteristics, and treatment modalities across included studies compromises the external validity and generalizability of the findings. Third, evidence supporting Do-IMRT and proton therapy relies on small-scale, single-institution studies, lacking robust data from large RCTs to confirm clinical efficacy and long-term outcomes. Fourth, imaging assessments, including MRI and CT, used to evaluate dysphagia-associated structural changes, are susceptible to interobserver variability, potentially affecting measurement accuracy. Finally, existing evidence regarding rehabilitation interventions for managing radiation-induced dysphagia in head and neck cancer predominantly comprises observational studies, with a scarcity of prospective RCTs to substantiate efficacy. To overcome these limitations, it is imperative that future research adopts standardized methodologies, undertakes extensive multicenter prospective studies, and implements rigorous RCTs. Such initiatives are anticipated to significantly improve the reliability, validity, and clinical utility of therapeutic strategies aimed at this patient population.
This review underscores advances in managing radiation-induced dysphagia in head and neck cancer. Dysphagia results from fibrosis and neuromuscular damage to pharyngeal constrictors and larynx. MRI and CT guide dose optimization, while Do-IMRT reduces exposure to these structures, enhancing swallowing outcomes. Early rehabilitation strengthens function, highlighting multidisciplinary care’s role. Balancing tumor control with swallowing preservation remains challenging. Future research should explore proton therapy and de-intensification protocols through RCTs to reduce morbidity. Continued innovation in imaging, dosimetry, and rehabilitation is essential to improve quality of life for survivors, refining clinical approaches to this persistent complication.
Received date: November 17, 2024
Accepted date: March 04, 2025
Published date: April 03, 2025
The manuscript has not been presented or discussed at any scientific meetings, conferences, or seminars related to the topic of the research.
This article is a narrative review based exclusively on previously published literature and publicly available data. It does not involve any new studies with human participants or animals performed by the authors. Therefore, ethical approval and informed consent were not required. The review adheres to the ethical principles outlined in the 1964 Helsinki Declaration and its subsequent amendments or comparable ethical standards.
The images included in this article were obtained from de-identified clinical imaging data and do not contain any personally identifiable information. Given that the images are anonymized and do not reveal patient identity, specific informed consent was not required. The study adheres to institutional and journal ethical guidelines regarding the use of clinical images for academic and research purposes. The authors affirm that all data presented comply with the ethical and legal requirements for scientific publication.
The author(s) of this research wish to declare that the study was conducted without the support of any specific grant from any funding agency in the public, commercial, or not-for-profit sectors. The author(s) conducted the study solely with their own resources, without any external financial assistance. The lack of financial support from external sources does not in any way impact the integrity or quality of the research presented in this article. The author(s) have ensured that the study was conducted according to the highest ethical and scientific standards.
In accordance with the ethical standards set forth by the SciTeMed publishing group for the publication of high-quality scientific research, the author(s) of this article declare that there are no financial or other conflicts of interest that could potentially impact the integrity of the research presented. Additionally, the author(s) affirm that this work is solely the intellectual property of the author(s), and no other individuals or entities have substantially contributed to its content or findings.
It is imperative to acknowledge that the opinions and statements articulated in this article are the exclusive responsibility of the author(s), and do not necessarily reflect the views or opinions of their affiliated institutions, the publishing house, editors, or other reviewers. Furthermore, the publisher does not endorse or guarantee the accuracy of any statements made by the manufacturer(s) or author(s). These disclaimers emphasize the importance of respecting the author(s)’ autonomy and the ability to express their own opinions regarding the subject matter, as well as those readers should exercise their own discretion in understanding the information provided. The position of the author(s) as well as their level of expertise in the subject area must be discerned, while also exercising critical thinking skills to arrive at an independent conclusion. As such, it is essential to approach the information in this article with an open mind and a discerning outlook.
© 2025 The Author(s). The article presented here is openly accessible under the terms of the Creative Commons Attribution 4.0 International License (CC-BY). This license grants the right for the material to be used, distributed, and reproduced in any way by anyone, provided that the original author(s), copyright holder(s), and the journal of publication are properly credited and cited as the source of the material. We follow accepted academic practices to ensure that proper credit is given to the original author(s) and the copyright holder(s), and that the original publication in this journal is cited accurately. Any use, distribution, or reproduction of the material must be consistent with the terms and conditions of the CC-BY license, and must not be compiled, distributed, or reproduced in a manner that is inconsistent with these terms and conditions. We encourage the use and dissemination of this material in a manner that respects and acknowledges the intellectual property rights of the original author(s) and copyright holder(s), and the importance of proper citation and attribution in academic publishing.
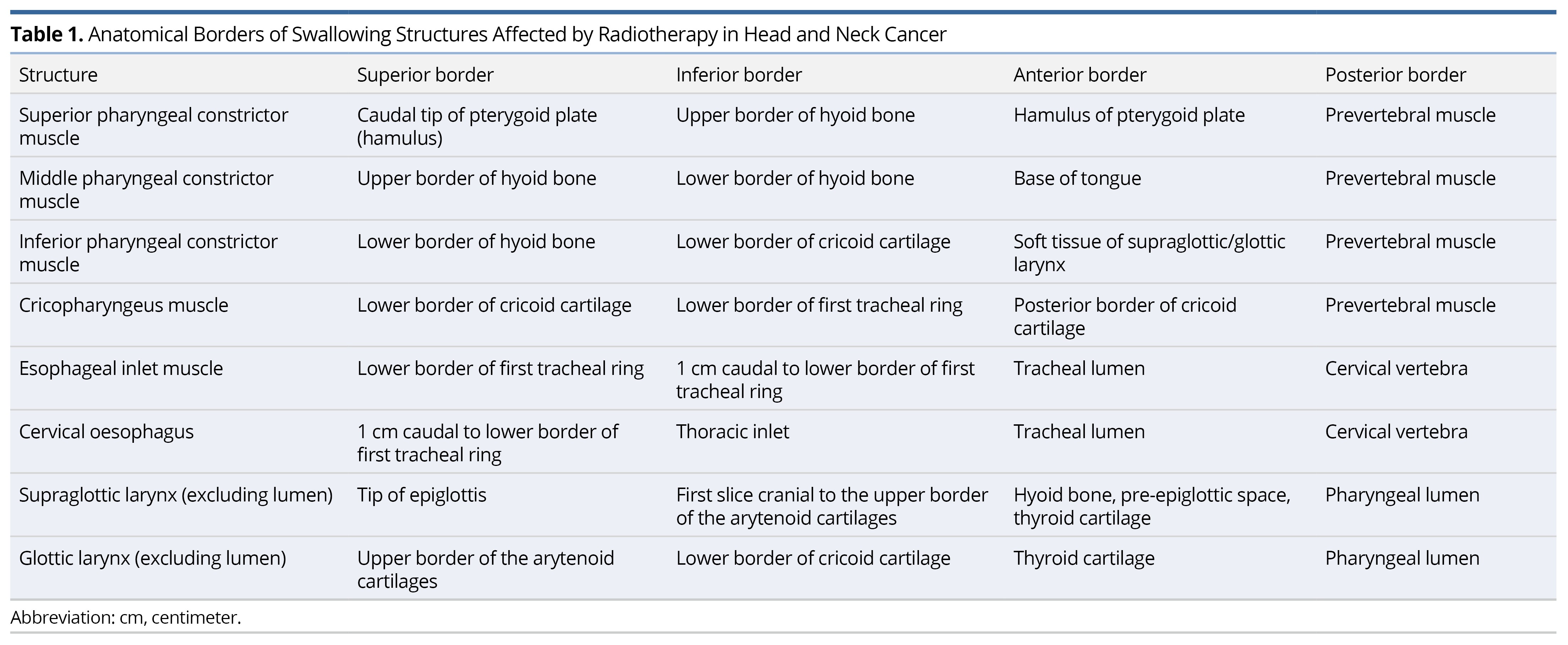
Radiation forms a vital part of neoadjuvant treatment in locally advanced rectal cancer (LARC) and recurrent rectal cancers. The adverse effects of radiation are well recognized; however, radiation-induced perforation at the tumour site is very rare and is poorly understood. A symptomatic rectal perforation requires an emergency surgical intervention. However, it may present silently and can give rise to suspicion of disease progression and/or residual disease on imaging. Authors present two cases of silent perforations. Both gave rise to a considerable diagnostic dilemma, which was resolved by careful evaluation with MRI.
The article under review contributes significantly to oncology by highlighting the impact of dysphagia-optimized intensity-modulated radiotherapy (DO-IMRT) in treating head and neck cancer patients. It focuses on enhancing the quality of life, extending beyond traditional treatment efficacy to fulfill the urgent need for patient-centered care. This research is set to profoundly influence clinical practices, guiding oncologists, radiologists, and care teams in refining therapeutic strategies for better patient outcomes. However, for acceptance in the International Microsurgery Journal, the authors are advised to revise the manuscript based on the reviewer and editorial comments. These revisions should include updating the literature review to incorporate the latest advancements up to 2024, where applicable, clarifying the research objectives to ensure they are explicitly defined, and providing clear definitions for technical terms such as Dmax, Dmean, and V50 to enhance readability and comprehension. Additionally, a descriptive title should be added to the table to provide context and improve its overall clarity. Addressing these points will align the article with the rigorous standards of our readership.
RevisionThe authors have carefully revised the manuscript in accordance with the reviewer’s, editor’s, and editorial team’s comments. To enhance the quality of the manuscript, the literature review has been updated to reflect recent advancements through 2024, particularly in imaging and radiotherapy techniques. The research objective has been clearly stated at the end of the Introduction to provide clear direction. Definitions for key dosimetric terms such as Dmax, Dmean, and V50 have been added at their first mention to improve clarity. Additionally, a descriptive title has been included for the table to enhance context and readability. These revisions collectively improve the manuscript’s structure, precision, and clinical relevance.
The authors explore the management of dysphagia in head and neck cancer patients using dysphagia-optimized intensity-modulated radiotherapy (DO-IMRT) in their article. This topic holds considerable clinical significance. Despite this, the article currently has shortcomings that compromise its readiness for publication. The literature review, crucial for grounding the study in the latest research, only extends to 2022. It does not include key studies or technological advancements from the last two years, which are vital given the rapid developments in this field. Additionally, the study’s objectives are too vague and fail to propose specific new research directions or address unresolved issues in the field. Addressing these critical gaps in content depth and scope is essential to elevate the article to the rigorous standards necessary for publication.
ResponseThe results of the DARS trial, published in Lancet Oncology in 2023, have been updated and incorporated into the manuscript under the section titled “Can Intensity-Modulated Radiotherapy (IMRT) Reduce Chemo-Radiotherapy–Associated Dysphagia?” The objective of this review is to provide a comprehensive overview of dysphagia following radiotherapy in head and neck cancers, a condition that is debilitating not only for patients but also for treating physicians and caregivers. The manuscript is intended to offer a holistic understanding of dysphagia for oncologists, radiologists, pathologists, and caregivers, with the aim of improving dysphagia-related quality of life in patients who have undergone radiotherapy for head and neck cancer.
This article examines the nuanced challenges of managing dysphagia in patients undergoing radical radiotherapy for head and neck cancers, highlighting the innovative potential of dysphagia-optimized intensity-modulated radiotherapy (DO-IMRT). It shifts the focus from traditional treatment efficacy to holistic quality of life impacts, emphasizing the importance of patient dignity and well-being in clinical practices. The insights provided are crucial for oncologists, radiologists, and supportive care teams committed to refining therapeutic strategies and enhancing patient experiences. Engaging and emotionally resonant, this article not only educates but also connects deeply with readers, making it a vital addition to contemporary medical literature. This article demonstrates expertise in its execution and merits publication.
The authors have meticulously addressed all reviewer and editorial feedback, demonstrating a commitment to scientific rigor and clarity. Reviewer 2’s concerns regarding outdated references and imprecise study objectives have been thoroughly resolved. The manuscript now incorporates the latest DARS trial findings (Lancet Oncology, 2023); however, Reference 31, originally cited from BMC Cancer (2016), remains outdated and should be updated to the 2023 Lancet Oncology publication. Additionally, while the refined manuscript scope, highlighting its clinical relevance to oncologists, radiologists, and caregivers, has been effectively articulated in the response to reviewers, this critical clarification has not yet been integrated into the final paragraph of the Introduction section. With these final adjustments, the manuscript meets the highest standards of scientific discourse, technical accuracy, and clinical applicability. Once the remaining minor revisions are implemented, this study will be well-positioned for publication.
ResponseReference 31 has been removed and replaced with the 2023 Lancet Oncology publication, with all subsequent references updated accordingly in both the manuscript and the reference list. Additionally, a critical clarification regarding the scope of the manuscript has been added to the final paragraph of the Introduction section.
Kundu S, Pramanik B, Mallik C, Majumder K, Ray A. Optimizing dysphagia management in head and neck cancer radiotherapy: Advanced imaging, precision dosimetry, and rehabilitation. Arch Otorhinolaryngol Head Neck Surg 2025;9(1):1. https://doi.org/10.24983/scitemed.aohns.2025.00194